Abstract
Fusarium pathogens are causal agents of several crop diseases and produce harmful mycotoxins resulting in crop and yield reduction worldwide. Among crop diseases, Fusarium wilt, Fusarium head blight, and Fusarium root blight are mostly reported diseases in numerous vegetables, crops, and fruits and have posed pressure on current food production and safety. In addition, the production of mycotoxins further aggravates plant health and causes serious health risks in humans and animals through food chain contamination. Different management practices have been enlisted in this chapter to reduce or eradicate Fusarium wilt in different crops. Interestingly, various mechanisms developed by plants have also been highlighted to fight against Fusarium pathogens and limit the growth of mycotoxins. One of defence mechanisms is plant antioxidant mechanisms to reduce oxidative stress by increasing enzymatic and non-enzymatic antioxidants to maintain cellular homeostasis under Fusarium infection. The other defence response is through hormonal signalling to combat fungal pathogens. Different phytohormones such as salicylic acid, ethylene, jasmonate, abscisic acid, cytokinin, auxin, and other plant secondary metabolites play a crucial part in the reduction of Fusarium growth and inhibit mycotoxin production through defence-related genes. Further, the use of different pre-harvest and post-harvest strategies has been elucidated to enhance plant resistance and growth by decreasing fungal pathogenicity and virulence.
Keywords
- antioxidants
- crop protection
- Fusarium pathogens
- mycotoxins
- phytohormones
1. Introduction
World’s population is growing exponentially and will reach 10 billion by 2050, adding pressure on available food production, its demand, and safety [1]. On the contrary, different environmental factors such as climate change, diseases, pests, and droughts are severely affecting current food supply and production system [2]. Different plant diseases cause crop reduction and yield losses which are major threats to food security globally. These diseases caused by various plant pathogens such as bacteria, fungi, and viruses can pose severe plant damage leading to economic losses [3, 4]. Among plant pathogens,
Plants are equipped with various plant defence mechanisms to combat these phytopathogens, and these mechanisms include plant physical barriers (structural changes), changes in different metabolic pathways to produce plant secondary metabolites (chemical changes), plant antioxidant system, hormonal signalling, and expression of defence-related genes [15, 16, 17]. Therefore, to prevent
1.1 Fusarium species as plant pathogens
These pathogenic species may enter into plant roots from soil or other media such as air, ground, and water. In addition, other factors including insects, injuries caused by new roots, and nematodes can lead to disease development or its symptoms such as chlorosis, necrosis, and wilting (Figure 1) [28]. The direct attack of
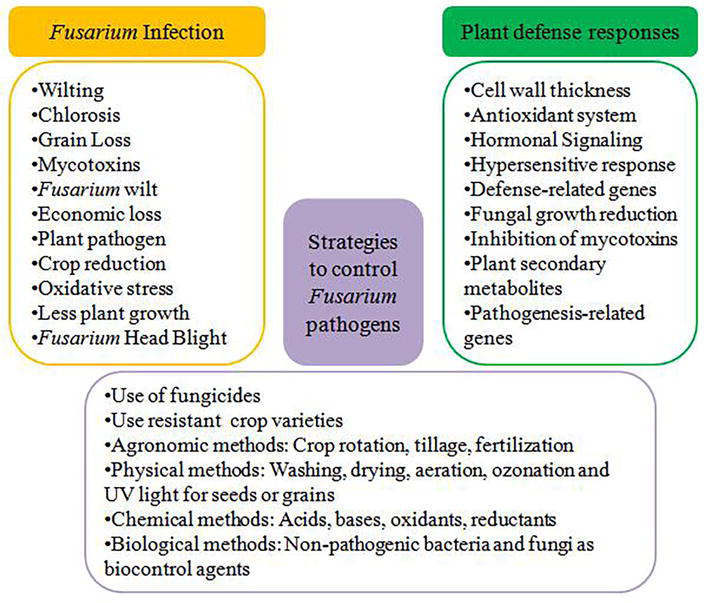
Figure 1.
Various effects caused by
2. Pathogen perception and plant defence system
Plants have evolved specific and effective defence systems to recognize
3. Host perception and Fusarium virulence
4. Production of Fusarium mycotoxins during plant-pathogen interaction
Fumonisins are polyketide-derived mycotoxins produced by various pathogens such as
Fusaric acid can also cause phytotoxic effects and result in wilt symptoms such as tomato wilt caused by
5. How plants deal with Fusarium mycotoxins
Plants employ mechanisms such as chemical modification and compartmentation of
In addition, genetic modification either by plant breeding or transgenesis can significantly prevent pathogens and their mycotoxins. For instance, rice plants expressing the
6. Fusarium wilt and its management
7. Plant antioxidant system to combat fungal pathogens
Guaiacol is a natural antioxidant and antifungal agent and has been studied against
8. Role of secondary metabolites in plant resistance
Various plant secondary metabolites are produced upon pathogenic attack or mycotoxin production. These secondary metabolites are involved not only in killing
9. Phytohormonal Defence Signalling against Fusarium Pathogens
Plant hormones play a crucial role in defence mechanisms to protect plants from
SA production is linked with
JA also plays an important part in signal transduction to induce plant defence responses under biotic stress conditions and is also involved in SAR during plant-pathogen interactions. For instance, JA treatment in rice significantly increased plant resistance against necrotrophic pathogens by activating the phenylpropanoid pathway [116]. The important roles of JA, SA, and ET were studied in tomato plants using gene markers of the hormones against
ET is a light gas molecule and is involved in the regulation of PTI to prevent fungal pathogens [120]. The ETR receptor (ETR1) is required by
10. Different strategies to control Fusarium pathogens
Plants employ different strategies to control
10.1 Biocontrol using bacterial species
Different species of
10.2 Biocontrol using fungal species
Non-pathogenic
10.3 Use of natural products from medicinal plants
Plant products exhibit antifungal properties and are eco-friendly in nature as these compounds do not persist in the environment for a longer period than other synthetic fungicides [143]. These plant extracts are widely being used in the agriculture sector to prevent fungal pathogens and control plant diseases. The antimicrobial property is based on the presence of certain secondary metabolites such as flavonoids, phenols, glycosides, alkaloids, polyphenols, tannins, and other compounds [144]. Numerous studies have elucidated the roles of plant products to prevent fungal infection of various
10.4 Pre- and post-harvest strategies to control Fusarium pathogens
The purpose of these preventive measures is to reduce
11. Future challenges
Numerous crops are susceptible to
12. Conclusion and future perspectives
It is concluded that
References
- 1.
FAO, IFAD, UNICEF, WFP, WHO. The State of Food Security and Nutrition in the World. Building Climate Resilience for Food Security and Nutrition. Rome. Italy: FAO; 2018 - 2.
Godfray HCJ, Beddington JR, Crute IR, Haddad L, Lawrence D, Muir JF, et al. Food security: The challenge of feeding 9 billion people. Science. 2010; 327 :812-818 - 3.
Cao Y, Zhang Z, Ling N, Yuan Y, Zheng X, Shen B, et al. Bacillus subtilis SQR 9 can controlFusarium wilt in cucumber by colonizing plant roots. Biology and Fertility of Soils. 2011;47 :495-506 - 4.
Mazzola M, Freilich S. Prospects for biological soil borne disease control: Application of indigenous versus synthetic microbiomes. Phytopathology. 2017; 107 :256-263 - 5.
Duan C, Qin Z, Yang Z, Li W, Sun S, Zhu Z, et al. Identification of pathogenic Fusarium spp. causing maize ear rot and potential mycotoxin production in China. Toxins. 2016;8 :186 - 6.
McGovern RJ. Management of tomato diseases caused by Fusarium oxysporum . Crop Protection. 2015;73 :78-92 - 7.
Gu Q , Yang Y, Yuan Q , Shi G, Wu L, Lou Z, et al. Bacillomycin D produced by bacillus amyloliquefaciens is involved in the antagonistic interaction with the plant-pathogenic fungusFusarium graminearum . Applied and Environmental Microbiology. 2017;83 :e01075-e01017 - 8.
Garcia D, Barros G, Chulze S, Ramos AJ, Sanchis V, Marín S. Impact of cycling temperatures on Fusarium verticillioides andFusarium graminearum growth and mycotoxins production in soybean. Journal of the Science of Food and Agriculture. 2012;92 :2952-2959 - 9.
Tekle S, Skinnes H, Bjørnstad A. The germination problem of oat seed lots affected by Fusarium head blight. European Journal of Plant Pathology. 2013;135 :147-158 - 10.
Hofgaard IS, Aamot HU, Torp T, Jestoi M, Lattanzio VMT, Klemsdal SS, et al. Associations between Fusarium species and mycotoxins in oats and spring wheat from farmers’ fields in Norway over a six-year period. World Mycotoxin Journal. 2016;9 :365-378 - 11.
Hjelkrem AGR, Torp T, Brodal G, Aamot HU, Strand E, Nordskog B, et al. DON content in oat grains in Norway related to weather conditions at different growth stages. European Journal of Plant Pathology. 2017; 148 :577-594 - 12.
Packa D, Załuski D, Graban Ł, Lajszner W, Hościk M. Reakcja diploidalnych, tetraploidalnych I heksaploidalnych pszenic na inokulację Fusarium culmorum (W.G.Smith) Sacc. Polish Journal of Agronomy. 2013;12 :38-48 - 13.
Salgado JD, Madden LV, Paul PA. Efficacy and economics of integrating in-field and harvesting strategies to manage Fusarium head blight of wheat. Plant Disease. 2014;98 :1407-1421 - 14.
Mielniczuk E, Skwaryło-Bednarz B. Fusarium head blight, mycotoxins and strategies for their reduction. Agronomy. 2020;10 (4):509 - 15.
Iqbal N, Czékus Z, Poór P, Ördög A. Plant defence mechanisms against mycotoxin Fumonisin B1. Chemico-Biological Interactions. 2021; 343 :109494 - 16.
Iqbal N, Czékus Z, Ördög A, Poór P. The main fungal pathogens and defence-related hormonal signalling in crops. In: Khan MIR, Singh A, Poór P, editors. Plant Hormones in Crop Improvement. Cambridge Academic Press; 2023. pp. 307-331 - 17.
Sadeghpour N, Asadi-Gharneh HA, Nasr-Esfahani M, Khankahdani HH, Golabadi M. Antioxidant enzymes associated with resistance to Fusarium oxysporum f. sp. melonis race 1.2 in melon. Physiological and Molecular Plant Pathology. 2022;121 :101880 - 18.
Seiber JN, Coats J, Duke SO, Gross AD. Biopesticides: State of the art and future opportunities. Journal of Agricultural and Food Chemistry. 2014; 62 :11613-11619 - 19.
Veliz EA, Martinez-Hidalgo P, Hirsch AM. Chitinase producing bacteria and their role in biocontrol. AIMS Microbiology. 2017; 3 :689-705 - 20.
Babadoost M. Fusarium: Historical and continued importance. In: Fusarium AT, editor. Plant Diseases, Pathogen Diversity, Genetic Diversity, Resistance and Molecular Markers. London, UK: Intech Open; 2018 - 21.
Mohamed Zubi WS, Mohd MH, Mohamed Nor NMI, Zakaria L. Fusarium species in mangrove soil in northern peninsular Malaysia and the soil physico-chemical properties. Microorganisms. 2021;9 :497 - 22.
Summerell BA, Laurence MH, Liew ECY, Leslie JF. Biogeography and phylogeography of Fusarium : A review. Fungal Diversity. 2010;44 :3-13 - 23.
Summerell BA, Leslie JF, Liew ECY, Laurence MH, Bullock S, Petrovic T, et al. Fusarium species associated with plants in Australia. Fungal Diversity. 2011;46 :1-27 - 24.
Iqbal N, Czékus Z, Ördög A, Poór P. Fusaric acid-evoked oxidative stress affects plant defence system by inducing biochemical changes at subcellular level. Plant Cell Reports. 2024; 43 (1):2 - 25.
Desai S, Dubey SC, Prasad RD. Impacts of climate change on Fusarium species Vis-à-Vis adaptation strategies. Indian Phytopathology. 2020;73 (4):593-603 - 26.
Iqbal N, Czékus Z, Angeli C, Bartók T, Poór P, Ördög A. Fumonisin B1-induced oxidative burst perturbed photosynthetic activity and affected antioxidant enzymatic response in tomato plants in ethylene-dependent manner. Journal of Plant Growth Regulation. 2023; 42 (3):1865-1878 - 27.
Rampersad SN. Pathogenomics and management of Fusarium diseases in plants. Pathogens. 2020;9 (5):340 - 28.
Incremona ME, Gonzalez M, Pioli RN, Salinas A. Infection of maize silks by a native Fusarium (Fusarium graminearum ) isolate in Argentina. Chilean Journal of Agricultural and Animal Science. 2014;30 :203-211 - 29.
Perincherry L, Lalak-Kańczugowska J, Stępień Ł. Fusarium -produced mycotoxins in plant-pathogen interactions. Toxins. 2019;11 (11):664 - 30.
Ekwomadu TI, Akinola SA, Mwanza M. Fusarium mycotoxins, their metabolites (free, emerging, and masked), food safety concerns, and health impacts. International Journal of Environmental Research and Public Health. 2021;18 (22):11741 - 31.
Eskola M, Kos G, Elliott CT, Hajšlová J, Mayar S, Krska R. Worldwide contamination of food-crops with mycotoxins: Validity of the widely cited ‘FAO estimate’ of 25%. Critical Reviews in Food Science and Nutrition. 2020; 60 :2773-2789 - 32.
Reddy KRN, Nurdijati SB, Salleh B. An overview of plant-derived products on control of mycotoxigenic fungi and mycotoxins. Asian Journal of Plant Sciences. 2010; 9 :126 - 33.
Nag P, Paul S, Shriti S, Das S. Defence response in plants and animals against a common fungal pathogen, Fusarium oxysporum . Current Research in Microbial Sciences. 2022;3 :100135 - 34.
Li CY, Deng GM, Yang J, Viljoen A, Jin Y, Kuang RB, et al. Transcriptome profiling of resistant and susceptible Cavendish banana roots following inoculation with Fusarium oxysporum f. sp. cubense tropical race 4. BMC Genomics. 2012;13 (1):1-11 - 35.
Muthamilarasan M, Prasad M. Plant innate immunity: An updated insight into defence mechanism. Journal of Biosciences. 2013; 38 :433-449 - 36.
Wojtasik W, Preisner M, Boba A, Kostyn K, Dymińska L, Hanuza J, et al. Rearrangement of cell wall polymers in flax infected with a pathogenic strain of Fusarium culmorum . Physiological and Molecular Plant Pathology. 2020;110 :101461 - 37.
Anisimova OK, Shchennikova AV, Kochieva EZ, Filyushin MA. Pathogenesis-related genes of PR1, PR2, PR4, and PR5 families are involved in the response to Fusarium infection in garlic (Allium sativum L.). International Journal of Molecular Sciences. 2021;22 (13):6688 - 38.
Beccari G, Hao G, Liu H. Fusarium pathogenesis: Infection mechanisms and disease progression in host plants. Frontiers in Plant Science. 2022;13 :1020404 - 39.
Husaini AM, Sakina A, Cambay SR. Host–pathogen interaction in Fusarium oxysporum infections: Where do we stand? Molecular Plant-Microbe Interactions. 2018;31 (9):889-898 - 40.
Ridenour JB, Bluhm BH. The HAP complex in Fusarium verticillioides is a key regulator of growth, morphogenesis, secondary metabolism, and pathogenesis. Fungal Genetics and Biology. 2014;69 :52-64 - 41.
Li M, Xie L, Wang M, Lin Y, Zhong J, Zhang Y, et al. FoQDE2-dependent milRNA promotes Fusarium oxysporum f. sp. cubense virulence by silencing a glycosyl hydrolase coding gene expression. PLoS Pathogens. 2022;18 (5):e1010157 - 42.
Perez-Nadales E, Di Pietro A. The transmembrane protein Sho1 cooperates with the mucin Msb2 to regulate invasive growth and plant infection in Fusarium oxysporum . Molecular Plant Pathology. 2015;16 (6):593-603 - 43.
Ploetz RC. Fusarium wilt of banana. Phytopathology. 2015;105 :1512-1521 - 44.
Tiwari N, Ahmed S, Kumar S, Sarker A. Fusarium wilt: A killer disease of lentil. In: Asku T, editor.Fusarium -Plant Diseases, Pathogen Diversity, Genetic Diversity, Resistance and Molecular Markers. London, UK, Rijeka, Croatia: IntechOpen; 2018 - 45.
Popescu S, Boldura OM, Borozan A, Madosa E. Molecular detection of Fusarium infections in wheat: A measure of quality assessment. Journal on Processing and Energy in Agriculture. 2022;26 (1):27-29 - 46.
Awuchi CG, Ondari EN, Eseoghene IJ, Twinomuhwezi H, Amagwula IO, Morya S. Fungal growth and mycotoxins production: Types, toxicities, control strategies, and detoxification. In: Sultan S, GKS S, editors. Fungal Reproduction and Growth. London: IntechOpen; 2021. p. 100207 - 47.
Stępień Ł, Koczyk G, Waśkiewicz A. FUM cluster divergence in fumonisins-producing Fusarium species. Fungal Biology. 2011;115 (2):112-123 - 48.
Gelineau-van WJ. Fumonisins. In: Gupta RC, editor. Reproductive and Developmental Toxicology. Cambridge: Academic Press; 2022. pp. 955-981 - 49.
Tiwari R, Shukla SK, Jaiswal VP, Tiwari RK. Pokkah boeng disease of sugarcane: Current status and opportunities. 2020; 12 (1):1-6 - 50.
Fernandes LB, Ghag SB. Molecular insights into the jasmonate signalling and associated defence responses against wilt caused by Fusarium oxysporum . Plant Physiology and Biochemistry. 2022;174 :22-34 - 51.
Niehaus EM, Díaz-Sánchez V, von Bargen KW, Kleigrewe K, Humpf HU, Limón MC, et al. Fusarins and fusaric acid in fusaria. In: Martín JF, García-Estrada C, Zeilinger S, editors. Biosynthesis and Molecular Genetics of Fungal Secondary Metabolites. New York: Springer; 2014. pp. 239-262 - 52.
Stępień Ł, Lalak-Kańczugowska J, Witaszak N, Urbaniak M. Fusarium secondary metabolism biosynthetic pathways: So close but so far away. In: Mérillon JM, Ramawat KG, editors. Co-Evolution of Secondary Metabolites. Cham: Springer Nature; 2020. pp. 211-247 - 53.
Selim ME, El-Gammal NA. Role of fusaric acid mycotoxin in pathogensis process of tomato wilt disease caused by Fusarium oxysporum . Journal of Bioprocessing and Biotechniques. 2015;5 :1 - 54.
Singh VK, Upadhyay RS. Fusaric acid induced cell death and changes in oxidative metabolism of Solanum lycopersicum L. Botanical Studies. 2014;55 :66 - 55.
Iqbal N, Czékus Z, Ördög A, Poór P. Ethylene-dependent effects of fusaric acid on the photosynthetic activity of tomato plants. Photosynthetica. 2021; 59 (2):337-348 - 56.
Singh VK, Singh HB, Upadhyay RS. Role of fusaric acid in the development of ‘ Fusarium wilt’ symptoms in tomato: Physiological, biochemical and proteomic perspectives. Plant Physiology and Biochemistry. 2017;118 :320-332 - 57.
Bertuzzi T, Giorni P, Rastelli S, Vaccino P, Lanzanova C, Locatelli S. Co-occurrence of moniliformin and regulated Fusarium toxins in maize and wheat grown in Italy. Molecules. 2020;25 (10):2440 - 58.
Urbaniak M, Stepie’n Ł, Uhlig S. Evidence for naturally produced Beauvericins containing N -methyl-tyrosine inHypocreale s Fungi. Toxins. 2019;11 :182 - 59.
Mallebrera B, Prosperini A, Font G, Ruiz MJ. In vitro mechanisms of Beauvericin toxicity: A review. Food and Chemical Toxicology. 2018; 111 :537-545 - 60.
Poppenberger B, Berthiller F, Lucyshyn D, Sieberer T, Schuhmacher R, Krska R, et al. Detoxification of the Fusarium mycotoxin deoxynivalenol by a UDP-glucosyltransferase fromArabidopsis thaliana . Journal of Biological Chemistry. 2003;278 :47905-47914 - 61.
Boddu J, Cho S, Muehlbauer GJ. Transcriptome analysis of trichothecene-induced gene expression in barley. Molecular Plant-Microbe Interactions. 2007; 20 :1364-1375 - 62.
Huang J, Pang C, Fan S, Song M, Yu J, Wei H, et al. Genome-wide analysis of the family 1 glycosyltransferases in cotton. Molecular Genetics and Genomics. 2015; 290 :1805-1818 - 63.
ter Beek J, Guskov A, Slotboom DJ. Structural diversity of ABC transporters. Journal of General Physiology. 2014; 143 (4):419-435 - 64.
Van Schie CC, Takken FL. Susceptibility genes 101: How to be a good host. Annual Review of Phytopathology. 2014; 52 :551-581 - 65.
Pereyra MG, Sulyok M, Baralla V, Dalcero AM, Krska R, Chulze S, et al. Evaluation of zearalenone, α-zearalenol, β-zearalenol, zearalenone 4-sulfate and β-zearalenol 4-glucoside levels during the ensiling process. World Mycotoxin Journal. 2014; 7 (3):291-295 - 66.
Ohsato S, Ochiai-Fukuda T, Nishiuchi T, Takahashi-Ando N, Koizumi S, Hamamoto H, et al. Transgenic rice plants expressing trichothecene 3- O -acetyltransferase show resistance to theFusarium phytotoxin deoxynivalenol. Plant Cell Reports. 2007;26 :531-538 - 67.
Li X, Shin S, Heinen S, Dill-Macky R, Berthiller F, Nersesian N, et al. Transgenic wheat expressing a barley UDP-glucosyltransferase detoxifies deoxynivalenol and provides high levels of resistance to Fusarium graminearum . Molecular Plant-Microbe Interactions. 2015;28 :1237-1246 - 68.
Perochon A, Váry Z, Malla KB, Halford NG, Paul MJ, Doohan FM. The wheat SnRK1α family and its contribution to Fusarium toxin tolerance. Plant Science. 2019;288 :110217 - 69.
Arie T. Fusarium diseases of cultivated plants, control, diagnosis, and molecular and genetic studies. Journal of Pesticide Science. 2019;44 (4):275-281 - 70.
Zakaria L. Fusarium species associated with diseases of major tropical fruit crops. Horticulturae. 2023;9 (3):322 - 71.
Khan N, Maymon M, Hirsch AM. Combating Fusarium infection usingBacillus -based antimicrobials. Microorganisms. 2017;5 (4):75 - 72.
Fourie G, Steenkamp ET, Ploetz RC, Gordon TR, Viljoen A. Current status of the taxonomic position of Fusarium oxysporum formae specialis cubense within theFusarium oxysporum complex. Infection, Genetics and Evolution. 2011;11 :533-542 - 73.
De Sain M, Rep M. The role of pathogen-secreted proteins in fungal vascular wilt diseases. International Journal of Molecular Sciences. 2015; 16 :23970-23993 - 74.
Mendel Z, Protosav A, Sharon M, Zveibil A, Ben Yahuda S, O’Donnell K, et al. An Asian ambrosia beetle Euwallacea fornicatus and its novel symbiotic fungusFusarium sp. Pose a serious threat to Israeli avocado industry. Phytoparasitica. 2012;40 :235-238 - 75.
Yadeta KA, Thomma BP. The xylem as battleground for plant hosts and vascular wilt pathogens. Frontiers in Plant Science. 2013; 4 :97 - 76.
Kazan K, Gardiner DM. Transcriptomics of cereal– Fusarium graminearum interactions: What we have learned so far. Molecular Plant Pathology. 2018;19 (3):764-778 - 77.
Del Ponte EM, Valent B, Bergstrom GC. A special issue on Fusarium head blight and wheat blast. Tropical Plant Pathology. 2017;42 :143-145 - 78.
Dweba CC, Figlan S, Shimelis HA, Motaung TE, Sydenham S, Mwadzingeni L, et al. Fusarium head blight of wheat: Pathogenesis and control strategies. Crop Protection. 2017;91 :114-122 - 79.
Seo Y, Kim YH. Potential reasons for prevalence of Fusarium wilt in oriental melon in Korea. Plant Pathology Journal. 2017;33 :249-263 - 80.
Ma YT, Fan HF, Gao YQ , Li H, Zhang AL, Gao JM. Natural products as sources of new fungicides (I): Synthesis and antifungal activity of acetophenone derivatives against phytopathogenic fungi. Chemical Biology and Drug Research. 2013; 81 :545-552 - 81.
Everts KL, Himmelstein JC. Fusarium wilt of watermelon: Towards sustainable management of are-emerging plant disease. Crop Protection. 2015;73 :93-99 - 82.
Heydari A, Pessarakli M. A review on biological control of fungal plant pathogens using microbial antagonists. Journal of Biological Sciences. 2010; 10 :273-290 - 83.
Bakkera MG, Gloverb JD, Maib JG, Kinkela LL. Plant community effects on the diversity and pathogen suppressive activity of soil streptomycetes . Applied Soil Ecology. 2010;46 :35-42 - 84.
Dunlap CA, Schisler DA, Bowman MJ, Rooney AP. Genomic analysis of Bacillus subtilis OH 131.1 and co-culturing withCryptococcus flavescens for control ofFusarium head blight. Plant Gene. 2015;2 :1-9 - 85.
Zalila-Kolsi I, Mahmoud AB, Ali H, Sellami S, Nasfi Z, Tounsi S, et al. Antagonist effects of bacillus spp. strains againstFusarium graminearum for protection of durum wheat (Triticum turgidum L. sub sp.durum ). Microbiological Research. 2016;192 :148-158 - 86.
Atanasova-Penichon V, Barreau C, Richard-Forget F. Antioxidant secondary metabolites in cereals: Potential involvement in resistance to Fusarium and mycotoxin accumulation. Frontiers in Microbiology. 2016;7 :566 - 87.
Chrpová J, Orsák M, Martinek P, Lachman J, Trávníčková M. Potential role and involvement of antioxidants and other secondary metabolites of wheat in the infection process and resistance to Fusarium spp. Agronom. 2021;11 (11):2235 - 88.
Anthony KK, George DS, Baldev Singh HK, Fung SM, Santhirasegaram V, Razali Z, et al. Reactive oxygen species activity and antioxidant properties of Fusarium infected bananas. Journal of Phytopathology. 2017;165 (4):213-222 - 89.
Zehra A, Meena M, Dubey MK, Aamir M, Upadhyay RS. Synergistic effects of plant defence elicitors and Trichoderma harzianum on enhanced induction of antioxidant defence system in tomato againstFusarium wilt disease. Botanical Studies. 2017;58 (1):1-14 - 90.
Hashem A, Akhter A, Alqarawi AA, Singh G, Almutairi KF, Abd_Allah EF. Mycorrhizal fungi induced activation of tomato defence system mitigates Fusarium wilt stress. Saudi Journal of Biological Sciences. 2021;28 (10):5442-5450 - 91.
Gao T, Zhang Y, Shi J, Mohamed SR, Xu J, Liu X. The antioxidant guaiacol exerts fungicidal activity against fungal growth and deoxynivalenol production in Fusarium graminearum . Frontiers in Microbiology. 2021;12 :762844 - 92.
Sun S, Yang Z, Song Z, Wang N, Guo N, Niu J, et al. Silicon enhances plant resistance to Fusarium wilt by promoting antioxidant potential and photosynthetic capacity in cucumber (Cucumis sativus L.). Frontiers in Plant Science. 2022;13 :1011859 - 93.
Matić M, Vuković R, Vrandečić K, Štolfa Čamagajevac I, Ćosić J, Vuković A, et al. Oxidative status and antioxidative response to Fusarium attack and different nitrogen levels in winter wheat varieties. Plants. 2021;10 (4):611 - 94.
Abdelaziz AM, Attia MS, Salem MS, Refaay DA, Alhoqail WA, Senousy HH. Cyanobacteria -mediated immune responses in pepper plants againstFusarium wilt. Plants. 2022;11 (15):2049 - 95.
Batnini M, Lopez-Gomez M, Palma F, Haddoudi I, Kallala N, Zribi K, et al. Sinorhizobium spp inoculation alleviates the effect ofFusarium oxysporum onMedicago truncatula plants by increasing antioxidant capacity and sucrose accumulation. Applied Soil Ecology. 2020;150 :103458 - 96.
Balmer D, Flors V, Glauser G, Mauch-Mani B. Metabolomics of cereals under biotic stress: Current knowledge and techniques. Frontiers in Plant Science. 2013; 4 :82 - 97.
Picot A, Atanasova-Pénichon V, Pons S, Marchegay G, Barreau C, Pinson-Gadais L, et al. Maize kernel antioxidants and their potential involvement in Fusarium ear rot resistance. Journal of Agricultural and Food Chemistry. 2013;61 :3389-3395 - 98.
Etzerodt T, Gislum R, Laursen BB, Heinrichson K, Gregersen PL, Jørgensen LN, et al. Correlation of deoxynivalenol accumulation in Fusarium-infected winter and spring wheat cultivars with secondary metabolites at different growth stages. Journal of Agricultural and Food Chemistry. 2016; 64 :4545-4555 - 99.
Kumaraswamy GK, Kushalappa AC, Choo TM, Dion Y, Rioux S. Differential metabolic response of barley genotypes, varying in resistance, to trichothecene-producing and-nonproducing (tri5-) isolates of Fusarium graminearum . Plant Pathology. 2012;61 :509-521 - 100.
Ferruz E, Atanasova-Pénichon V, Bonnin-Verdal MN, Marchegay G, Pinson-Gadais L, Ducos C, et al. Effects of phenolic acids on the growth and production of T-2 and HT-2 toxins by Fusarium langsethiae andF. Sporotrichioides . Molecules. 2016;21 :449 - 101.
Katagiri F, Tsuda K. Understanding the plant immune system. Molecular Plant Microbe Interactions. 2010; 23 (12):15311536 - 102.
Couto D, Zipfel C. Regulation of pattern recognition receptor signalling in plants. Nature Reviews Immunology. 2016; 16 :537-552 - 103.
Verma V, Ravindran P, Kumar PP. Plant hormone-mediated regulation of stress responses. BMC Plant Biology. 2016; 16 (1):1-10 - 104.
Ghorbel M, Brini F. Hormone mediated cell signalling in plants under changing environmental stress. Plant Gene. 2021; 28 :100335 - 105.
Yu SG, Kim JH, Cho NH, Oh TR, Kim WT. Arabidopsis RING E3 ubiquitin ligase JUL1 participates in ABA-mediated microtubule depolymerization, stomatal closure, and tolerance response to drought stress. The Plant Journal. 2020;103 (2):824-842 - 106.
Torres-Vera R, García JM, Pozo MJ, López-Ráez JA. Do strigolactones contribute to plant defence? Molecular Plant Pathology. 2014; 15 (2):211-216 - 107.
Takatsuji H, Jiang CJ. Plant hormone crosstalks under biotic stresses. In: Tran LS, Pal S, editors. Phytohormones: A Window to Metabolism, Signalling and Biotechnological Applications. New York: Springer; 2014. pp. 323-350 - 108.
Qi PF, Johnston A, Balcerzak M, Rocheleau H, Harris LJ, Long XY, et al. Effect of salicylic acid on Fusarium graminearum , the major causal agent ofFusarium head blight in wheat. Fungal Biology. 2012;116 (3):413-426 - 109.
Makandar R, Nalam V, Chaturvedi R, Jeannotte R, Sparks AA, Shah J. Involvement of interaction between salicylic acid and jasmonate signalling pathways in Arabidopsis interaction withFusarium graminearum . Molecular Plant Microbe Interactions. 2010;23 :861-870 - 110.
Yang YX, J Ahammed G, Wu C, Fan SY, Zhou YH. Crosstalk among jasmonate, salicylate and ethylene signalling pathways in plant disease and immune responses. Current Protein and Peptide Science. 2015; 16 (5):450-461 - 111.
Xue RF, Wu J, Wang LF, Blair MW, Wang XM, De Ge W, et al. Salicylic acid enhances resistance to Fusarium oxysporum f. sp. phaseoli in common beans (Phaseolus vulgaris L .). Journal of Plant Growth Regulation. 2013;33 :470-476 - 112.
Djamei A, Schipper K, Rabe F, Ghosh A, Vincon V, Kahnt J, et al. Metabolic priming by a secreted fungal effector. Nature. 2011; 478 :395-398 - 113.
Han X, Kahmann R. Manipulation of phytohormone pathways by effectors of filamentous plant pathogens. Frontiers in Plant Science. 2019; 10 :822 - 114.
Chakraborty N. Salicylic acid and nitric oxide cross-talks to improve innate immunity and plant vigor in tomato against Fusarium oxysporum stress. Plant Cell Reports. 2021;40 (8):1415-1427 - 115.
Guan Y, He X, Wen D, Chen S, Chen F, Chen F, et al. Fusarium oxysporum infection on root elicit aboveground terpene production and salicylic acid accumulation inChrysanthemum morifolium . Plant Physiology and Biochemistry. 2022;190 :11-23 - 116.
Taheri P, Tarighi S. Riboflavin induces resistance in rice against Rhizoctonia solani via jasmonate-mediated priming of phenylpropanoid pathway. Journal of Plant Physiology. 2010;167 (3):201-208 - 117.
Di X, Gomila JO, Takken FL. Involvement of salicylic acid, ethylene and jasmonic acid signalling pathways in the susceptibility of tomato to Fusarium oxysporum . Molecular Plant Pathology. 2017;18 (7):1024-1035 - 118.
Qi PF, Balcerzak M, Rocheleau H, Leung W, Wei YM, Zheng YL, et al. Jasmonic acid and abscisic acid play important roles in host–pathogen interaction between Fusarium graminearum and wheat during the early stages ofFusarium head blight. Physiological and Molecular Plant Pathology. 2016;93 :39-48 - 119.
Siciliano I, Amaral Carneiro G, Spadaro D, Garibaldi A, Gullino ML. Jasmonic acid, abscisic acid, and salicylic acid are involved in the phytoalexin responses of rice to Fusarium fujikuroi , a high gibberellin producer pathogen. Journal of Agricultural and Food Chemistry. 2015;63 (37):8134-8142 - 120.
Anver S, Tsuda K. Ethylene and plant immunity. In: Wen CK, editor. Ethylene in Plants. Dordrecht: Springer; 2015. p. 205221 - 121.
Pantelides IS, Tjamos SE, Pappa S, Kargakis M, Paplomatas EJ. The ethylene receptor ETR1 is required for Fusarium oxysporum pathogenicity. Plant Pathology. 2013;62 :1302-1309 - 122.
Chen X, Steed A, Travella S, Keller B, Nicholson P. Fusarium graminearum exploits ethylene signalling to colonize dicotyledonous and monocotyledonous plants. New Phytologist. 2009;182 :975-983 - 123.
Dinolfo MI, Castañares E, Stenglein SA. Fusarium -plant interaction: State of the art-a review. Czech Academy of Agricultural Sciences; Plant Protection Science. 2017;53 (2):61-70 - 124.
Cakir B, Gül A, Yolageldi L, Özaktan H. Response to Fusarium oxysporum f.sp. radices-lycopersici in tomato roots involves regulation of SA- and ET-responsive gene expressions. European Journal of Plant Pathology. 2014;139 :379-391 - 125.
Luu VT, Schuck S, Kim SG, Weinhold A, Baldwin IT. Jasmonic acid signalling mediates resistance of the wild tobacco Nicotiana attenuata to its nativeFusarium , but noAlternaria , fungal pathogens. Plant, Cell and Environment. 2015;38 :572-584 - 126.
Asano T, Kimura M, Nishiuchi T. The defence response in Arabidopsis thaliana againstFusarium sporotrichioides . Proteome Science. 2012;10 :61 - 127.
Gao CS, Kou XJ, Li HP, Zhang JB, Saad ASI, Liao YC. Inverse effects of Arabidopsis NPR1 gene onFusarium seedling blight andFusarium head blight in transgenic wheat. Plant Pathology. 2012;62 :383-392 - 128.
Haidoulis JF, Nicholson P. Different effects of phytohormones on Fusarium head blight andFusarium root rot resistance inBrachypodium distachyon . Journal of Plant Interactions. 2020;15 (1):335-344 - 129.
Law JWF, Ser HL, Khan TM, Chuah LH, Pusparajah P, Chan KG, et al. The potential of Streptomyces as biocontrol agents against the rice blast fungus,Magnaporthe oryzae (Pyricularia oryzae ). Frontiers in Microbiology. 2017;8 :3 - 130.
Donato V, Ayala FR, Cogliati S, Bauman C, Costa JB, Lenini C, et al. Bacillus subtilis biofilm extendsCaenorhabditis elegans longevity through downregulation of the insulin-like signalling pathway. Nature Communications. 2017;8 :14332 - 131.
Allard-Massicotte R, Tessier L, Lecuyer F, Lakshmanan V, Lucier JF, Garneau D, et al. Bacillus subtilis early colonization ofArabidopsis thaliana roots involve multiple chemotaxis receptors. mBio. 2016;7 :e01664-e01616 - 132.
Heidarzadeh N, Baghaee-Ravari S. Application of Bacillus pumilus as a potential biocontrol agent ofFusarium wilt of tomato. Archives of Phytopathology and Plant Protection. 2015;48 :13-16 - 133.
Swiontek BM, Jankiewicz U, Burkowska A, Walczak M. Chitinolytic microorganisms and their possible application in environmental protection. Current Microbiology. 2014; 68 :71-81 - 134.
Zhao Y, Selvaraj JN, Xing F, Zhou L, Wang Y, Song H, et al. Antagonistic action of Bacillus subtilis strain SG6 onFusarium graminearum . PLoS One. 2014;9 :e92486 - 135.
Gomaa EZ. Chitinase production by Bacillus thuringiensis andBacillus licheniformis: Their potential in antifungal biocontrol. Journal of Microbiology. 2012;50 :103-111 - 136.
Akram W, Anjum T, Ali B. Phenylacetic acid is ISR determinant produced by Bacillus fortis IAGS162, which involves extensive re-modulation in metabolomics of tomato to protect againstFusarium wilt. Frontiers in Plant Science. 2016;7 :498 - 137.
Patil S, Sriram S. Biological control of Fusarium wilt in crop plants using non-pathogenic isolates ofFusarium species. Indian Phytopathology. 2020;73 (1):11-19 - 138.
Thangavelu R, Mustaffa MM. Current advances in the Fusarium wilt disease management in banana with emphasis on biological control. In: Cumagun CJ, editor. Plant Pathology. London: IntechOpen; 2012. pp. 273-288 - 139.
Wojtasik W, Kulma A, Namys K, Preisner M, Szopa J. Polyamine metabolism in flax in response to treatment with pathogenic and non-pathogenic Fusarium strains. Frontiers in Plant Science. 2015;6 :291 - 140.
Abderrahmane S, Merzoug A, Taleb M. The use of non-pathogenic Fusarium oxysporum as a biocontrol againstFusarium wilt of date palm (Bayoud disease). In: Proceedings of International Workshop on “Sustainability of Phoenici” Culture Production Systems in Algeria. Biskra. Algeria: University of Biskra; 2016. p. 67 - 141.
Nawar LS. Phytochemical and SDS-dissociated proteins of pathogenic and non-pathogenic Fusarium oxysporum isolates. International Journal of ChemTech Researh. 2016;9 (6):165-172 - 142.
Podgórska-Kryszczuk I, Solarska E, Kordowska-Wiater M. Biological control of Fusarium culmorum ,Fusarium graminearum andFusarium poae by antagonistic yeasts. Pathogens. 2022;11 (1):86 - 143.
Seepe HA, Lodama KE, Sutherland R, Nxumalo W, Amoo SO. In vivo antifungal activity of south African medicinal plant extracts against Fusarium pathogens and their phytotoxicity evaluation. Plants. 2020;9 :1668 - 144.
Sultana S, Akhtar N, Asif HM. Phytochemical screening and antipyretic effects of hydro-methanol extract of Melia azedarach leaves in rabbits. Bangladesh Journal of Pharmacology. 2013;8 :214-217 - 145.
Seepe HA, Nxumalo W, Amoo SO. Natural products from medicinal plants against phytopathogenic Fusarium species: Current research endeavours, challenges and prospects. Molecules. 2021;26 (21):6539 - 146.
Mogashoa MM, Masoko P, Eloff JN. Different Combretum molle (Combretaceae) leaf extracts contain several different antifungal and antibacterial compounds. South African Journal of Botony. 2019;126 :322-327 - 147.
Seepe HA, Amoo SO, Nxumalo W, Adeleke RA. Antifungal activity of medicinal plant extracts for potential management of Fusarium pathogens. Research on Crops. 2019;20 :399-406 - 148.
Seepe HA, Amoo SO, Nxumalo W, Adeleke RA. Sustainable use of thirteen south African medicinal plants for the management of crop diseases caused by Fusarium species–An in vitro study. South African Journal of Botony. 2020;130 :456-464 - 149.
Li KM, Dong X, Ma YN, Wu ZN, Yan YM, Cheng YX. Antifungal coumarins and lignans from Artemisia annua . Fitoterapia. 2019;134 :323-328 - 150.
He R, Guo D, Huang Z, Kong Y, Ji C, Gu J, et al. Systematic investigation of stereochemistry, stereoselective bioactivity, and antifungal mechanism of chiral triazole fungicide metconazole. Science of the Total Environment. 2021; 784 :147194 - 151.
Solanki MK, Abdelfattah A, Britzi M, Zakin V, Wisniewski M, Droby S, et al. Shifts in the composition of the microbiota of stored wheat grains in response to fumigation. Frontiers in Microbiology. 2019; 10 :1098 - 152.
Juodeikiene G, Bartkiene E, Cernauskas D, Cizeikiene D, Zadeike D, Lele V, et al. Antifungal activity of lactic acid bacteria and their application for Fusarium mycotoxins reduction in malting wheat grains. LWT-Food Science Technology. 2018;89 :307-314 - 153.
Omotayo OP, Omotayo AO, Mwanza M, Babalola OO. Prevalence of mycotoxins and their consequences on human health. Toxicological Research. 2019; 35 :1-7