The percentage of each non-
Abstract
Wine can be regarded as a nutritional source for the human diet. It contains many nutrients such as vitamins and minerals, organic acids, flavonoids, and terpenoids. The varietal aroma of wines originated from the symbionts of the grapes and epiphytic microbiota, which combinedly grew from the sexual reproduction of the plant through seeds or during clonal reproduction. Nowadays, more and more studies focus on the assembly process of epiphytic microbiota and design a synthetic microbial community based on regional characteristics to improve wine quality and biofunctions. This review synthesizes the current concepts on the construction of synthetic microbiota, analyzes the advantages and difficulties in designing a functional yeast community, and lists the practical tools for data processing and model construction. A well-designed yeast community will possess high robustness against environment interference, higher efficiency of fermentation, and higher yield of targeted bioproducts.
Keywords
- non-Saccharomyces
- top-down strategy
- systematic biology
- untargeted metabolomics
- icewine
- spontaneous fermentation
1. Introduction
The application of commercial yeast in an incompatible fermentation scenario potentially causes the risk of stuck and sluggish fermentation and product homogenization. It even has a detrimental effect on the composition of regional microbiota [1], which play important roles in characterizing aroma profile and wine quality [2]. Currently, more and more researchers and wineries have realized the limitations of commercial yeasts and are gradually turning their attention to the study of non-
Icewine is a type of dessert wine produced from grape juice extracted from frozen grapes [11]. It is normally characterized by a fruity and flowery aroma and smooth taste. Icewine making is heavily dependent on the local environmental abiotic factors during late harvest. Fully ripe grapes hang on the vines for several months to suffer natural freeze-thaw cycles and be desiccated for the concentration of sugar (>35°Brix) [12]. This period has been proven to be effective for the accumulation of varietal aroma in grapes [2, 5]. For instance, high-odor-active compounds, such as terpenes and phenylalanine-derivates, can be largely developed during late harvest through a series of interactions between epiphytic microeukaryotes and grapes [2, 5]. Our previous studies have revealed the freeze-thaw cycles are the inevitable abiotic factors that cause cellular degradation and compartmentation of grape skin [2] and improve the aroma complexity of grapes during late harvest [5]. Therefore, selecting an icewine region is the essential prerequisite for producing high-quality icewine, which must present the regional characteristics [13]. Huanren area (Liaoning Province, Northeast China) is a representative icewine-producing area in China. It is characterized by a year-round cold climate, fewer problems of pests and disease [2]. Unique ecology shapes distinctive and stress-tolerant microeukaryotic communities, which remain active in pressed grape juice and become determinant to icewine fermentation [2, 13]. However, the mechanism and driving factors of the microeukaryotic assembly process are still unclear during icewine fermentation.
Metabolomics mainly studies the variety, quantity, and change rule of the metabolites with molecular weight less than 1500 Da caused by external stimulation, pathophysiological changes, and gene mutation. It is an extension of transcriptomics and proteomics, which accurately reflect the physiological state of organisms. Therefore, it effectively reveals the biological processes of biomarkers, the mechanism of the biological activities, and the regulatory pathways [14]. In terms of detection modes, metabolomics is mainly divided into untargeted analysis and targeted analysis. The untargeted metabolomics is usually based on a high-resolution mass spectrometer (triple TOF or QE) [15]. It can perform unbiased, large-scale, and systematic detection of various metabolites in experimental samples, providing an ‘aerial photography’ perspective to reflect the metabolic disturbance in the plants and microorganisms [16].
This study jointly used untargeted metabolomics and high-throughput sequencing technology to verify the effects of the self-assembled non-

Figure 1.
Four inoculation methods were carried out for icewine fermentation. CS and HS were inoculated using the epiphytic microorganisms from the fully ripe grape berries and the grape berries in late harvest, respectively. HN was inoculated using the six core epiphytic yeast strains, which were important microeukaryotes in the grape skin. Control was conducted in icewine fermentation using a commercial yeast strain (BV818). Untargeted metabolomics and high-throughput sequencing technology were jointly used to analyze the self-assembly process of micro eukaryotic communities and the driving factors (important metabolites) during icewine fermentation.
2. Materials and methods
2.1 Vinification of V. blanc
Spontaneous fermentation using the epiphytic microorganisms from the fully ripe grape berries (CS): The ripe
Spontaneous fermentation using the epiphytic microorganisms from the grape berries in late harvest (HS): based on previous research,
Icewine fermentation using the self-assembled non-
| No. | Yeast species | Importance | Initial ratio (%) | Inoculation (mL) |
|---|---|---|---|---|
| 1 | 88.05 | 21.7 | 8.7 | |
| 2 | 80.59 | 19.8 | 7.9 | |
| 3 | 77.00 | 18.9 | 7.6 | |
| 4 | 63.69 | 15.7 | 6.3 | |
| 5 | 57.59 | 14.2 | 5.7 | |
| 6 | 39.62 | 9.7 | 3.9 |
Table 1.
Icewine fermentation using commercial yeast as control (BV818): a commercial yeast BV818 (AngelYeast Co., Ltd., Yichang) was inoculated into the filtrated grape juice for starting AF as control. This yeast belongs to
2.2 Viticulture and grape juice filtration
2.3 Determination of physicochemical parameters of icewine
Basic wine physicochemical parameters, such as hue, color intensity, Brix°, total sugar, total acid, glucose/fructose, lactic acid, malic acid, acetic acid, and glycerol, were determined using Y15 enzymatic autoanalyzer (Biosystems S.A., Barcelona, Spain). These analyses were performed using the appropriate enzymatic reaction kits purchased from Biosystems. Before detection, the Y-15 was calibrated with external standards that were technically supported by the Biosystems enterprise (www.biosystems.es). The pH value was measured using a PB-10 pH meter (Sartorius, Göttingen, Germany). The alcoholic degree of icewine was determined based on the National Standards of P. R. China (GB/T 15038-2006) [2, 4].
2.4 Untargeted metabolomics analysis
2.4.1 Sample collection and preparation
Icewine samples were centrifuged (4°C, 10,000 rpm, 10 min). The supernatant was added into precooled methanol/acetonitrile/water solution (2:2:1, v/v). The mixture was carried out vortex, ultrasonic homogenization (4°C, 30 min), stewing (−20°C, 10 min), and centrifugation (4°C, 12,000 rpm, 20 min). Then, the supernatant was conducted vacuum freeze-drying. Before metabolomics analysis, 100 μL acetonitrile aqueous solution (acetonitrile:water = 1:1, v/v) was added to redissolve the dry sample. Subsequently, the solution was vortexed and centrifuged (4°C, 12,000 rpm, 15 min) for UHPLC-MS analysis. Twenty microliters of each sample were mixed as a QC sample [17].
2.4.2 UHPLC conditions
Analyses were performed using a UHPLC (1290 Infinity LC, Agilent Technologies) coupled to a quadrupole time-of-flight (AB Sciex Triple TOF 6600). For HILIC separation, samples were analyzed using a 2.1 mm × 100 mm ACQUIY UPLC BEH 1.7 μm column (waters, Ireland). In both ESI positive and negative modes, the mobile phase contained A = 25 mM ammonium acetate and 25 mM ammonium hydroxide in water, and B = acetonitrile. The gradient was 85% B for 1 min, linearly reduced to 65% in 11 min, then reduced to 40% in 0.1 min, kept for 4 min, and then increased to 85% in 0.1 min, with a 5 min re-equilibration period employed.
2.4.3 ESI-Q-TOF MS/MS conditions
The ESI source conditions were set as follows: Ion Source Gas1 (Gas1) as 60, Ion Source Gas2 (Gas2) as 60, curtain gas (CUR) as 30, source temperature: 600°C, IonSpray Voltage Floating (ISVF) ± 5500 V. In MS-only acquisition, the instrument was set to acquire over the
2.4.4 Data processing
The raw MS data (wiff.scan files) were converted to MzXML files using ProteoWizard MSConvert before importing them into freely available XCMS software. CAMERA (Collection of Algorithms of Metabolite Profile Annotation) was used for the annotation of isotopes and adducts [18]. In the extracted ion features, only the variables having more than 50% of the nonzero measurement values in at least one group were kept. Compound identification of metabolites was performed by comparing the accuracy
2.5 Metabarcoding of internal transcribed spacer (ITS) sequence
The quantity and quality of extracted DNA were measured using a NanoDrop ND-1000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA) and agarose gel electrophoresis, respectively. To analyze the taxonomic composition of the microeukaryotes communities, ITS1 of microeukaryotes 18S rRNA genes via a two-step amplification procedure using primers ITS1F (5′-CTTGGTCATTTAGAGGAAGTAA-3′), and ITS1R (5′-GCTGCGTTCTTCATCGATGC-3′) were carried out. Specific DNA extraction, PCR, and Illumina MiSeq sequencing (2- by 150-bp reads) were performed using the Illumina MiSeq platform at Shanghai Personal Biotechnology Co., Ltd. (Shanghai, China) [4]. Each sample was extracted for use in three replicates, and each extraction was analyzed twice. The Quantitative Insights into Microbial Ecology (QIIME2) pipeline was employed to process the sequencing data. Amplicon sequence variants were obtained using the procedure of denoise, quality control, splicing, and mosaicking, which were carried out using denoise-paired QIIME DADA2.
2.6 Statistical analysis
Statistics for the characteristic metabolites in
3. Results
3.1 Identification of the characteristic metabolites
The results of PCA and heatmap cluster of metabolomic illustrated different metabolite profiles between the four fermentation scenarios and

Figure 2.
Comparison of characteristic metabolites in the icewine fermentation and grape juice. (A) Heatmap cluster of all identified metabolites in the four icewine fermentation and grape juice, especially the high-valued terpenes and flavonoids, were calculated and highlighted on the left side of the heatmap diagram. The importance of each treatment was shown on the top of the heatmap diagram. Herein, the Euclidean method was used for distance measurement, and the clustering algorithm was Ward. (B) Score plot of PLS-DA for analysis of observation of four icewine fermentations. (C) Ranking of characteristic metabolites by weighting the sum of VIP scores in compound 1 of PLS-DA. Colored boxes on the right side indicated the correlations between each metabolite and fermentation. Note: CS: spontaneous fermentation using the epiphytic microorganisms from the fully ripe grape berries; HS: spontaneous fermentation using the epiphytic microorganisms from the grape berries in late harvest; HN: icewine fermentation using the self-assembled non-Saccharomyces yeasts group icewine fermentation using a commercial yeast strain, which belongs to
Partial least squares discrimination analysis (PLS-DA) was carried out to compare the differences in each fermentation scenario (Figure 2B). Component 1 and component 2 contributed 76.6% of PLS-DA in total. It indicated the metabolite profile of each fermentation can be discriminated with the first two components. The four fermentations showed a far distance from the grape juice. Herein, the spontaneous fermentations (HS and CS) were close to HN, while BV818 showed a different metabolite profile from the others. To study the characteristic metabolites and their correlations to each fermentation. The top 20 characteristic metabolites were ranked according to their VIP scores of PLS-DA (Figure 2C). Most characteristic metabolites in icewine were positively correlated to the spontaneous fermentation and HN except glycerophosphocholine, succinate, and neohesperidose. In terms of spontaneous fermentation, some characteristic metabolites commonly showed positive correlations to CS and HS, such as d-gluconate, trans-3′-hydroxycotinine o-β-d-glucuronide, d-galacturonic acid, 2-isopropylmalic acid, gramine, adenine, and two dipeptides (Leu-His and Thr-Leu).
3.2 Spatial dispersal of microeukaryotic communities and correlations to the characteristic metabolites during AF
The spatial dispersal and co-occurrence network of each fermentation scenario jointly showed the dynamic changes of microeukaryotes during AF (Figure 3). Accordingly, the microeukaryotes in BV818 were highly developed at the end stage of AF, where ASVs of the microeukaryotes showed a higher enrichment but lower diffusivity (Figure 3A). The inner interactions between microeukaryotes in each module were positive. The major microeukaryotic modules in BV818 were modules 1, 2, 9, 7, and 14. Herein, the cross interactions between modules 1, 2, 7, and 9 showed higher negative similarity. It indicated the major activities between these modules were dominated by negative cohesion, such as competition, parasitism, or amensalism [21]. Moreover, module 14 showed fewer interactions with other modules. It indicated the related microeukaryotic communities could independently develop and be neutralized to other species during AF. HN showed a contrary spatial dispersal but a similar co-occurrence network to BV818 (Figure 3C). The strongest diffusivity of microeukaryotes was observed at the beginning of AF, while the weakest diffusivity was presented at the end of AF. The major microeukaryotic modules of HN were 1, 2, 6, and 12. The cross interactions between modules 1, 2, and 6 were also dominated by negative cohesion; meanwhile, these modules showed fewer interactions with module 12. Both CS and HS belonging to spontaneous fermentation and the same microbial origination from a vineyard resulted in similar spatial dispersals and co-occurrence networks of microeukaryotes during AF (Figure 3B and D). ASVs of microeukaryotes were evenly distributed in all the stages of AF. Comparatively, the microeukaryotes of CS mainly developed at the beginning and end stages of AF, while the microeukaryotes’ development of HS was vigorous at the middle stage of AF. The spontaneous fermentation presented more modules and more complex interactions than BV818 and HN. The major microeukaryotic modules in CS were 1, 2, 6, 8, 10, and 15. Herein, module 1 and module 10 showed positive cross interactions, namely, cooperation and mutualism. However, other microeukaryotic modules were mainly controlled by negative cohesion. The major modules in HS were 1, 2, 6, 7, 9, and 10. Herein, module 1 and module 7 showed positive cross interactions, while other modules were controlled by negative cohesion. In general, the cross-interaction between microeukaryotic phyla during AF was dominated by negative cohesion, and the abundance patterns of microeukaryotic communities depend on different fermentation modes [22].

Figure 3.
(A) Spatial dispersal and co-occurrence networks of fungi genera of BV818 on three stages during icewine fermentation. (B) Spatial dispersal and co-occurrence networks of fungi genera of CS on three stages during icewine fermentation. (C) Spatial dispersal and co-occurrence networks of fungi genera of HN on three stages during icewine fermentation. (D) Spatial dispersal and co-occurrence networks of fungi genera of HS on three stages during icewine fermentation. Each node represents one ASV that is labeled by genus. A node was verified by a robust (Spearman’s correction coefficient R > 0.6) and significant (pFDR < 0.05) correlation. The size of each node is relational to the number of connections, while nodes with the same color display the same module. The thickness of each connection between two nodes is relational to the strength of Spearman’s correlation coefficient. Note: CS: spontaneous fermentation using the epiphytic microorganisms from the fully ripe grape berries; HS: spontaneous fermentation using the epiphytic microorganisms from the grape berries in late harvest; HN: icewine fermentation using the self-assembled non-Saccharomyces yeasts group, icewine fermentation using a commercial yeast strain, which belongs to
Mantel test was used to study the correlations between the specific order of microeukaryotes and fermentation mode (Figure 4A). As a result,
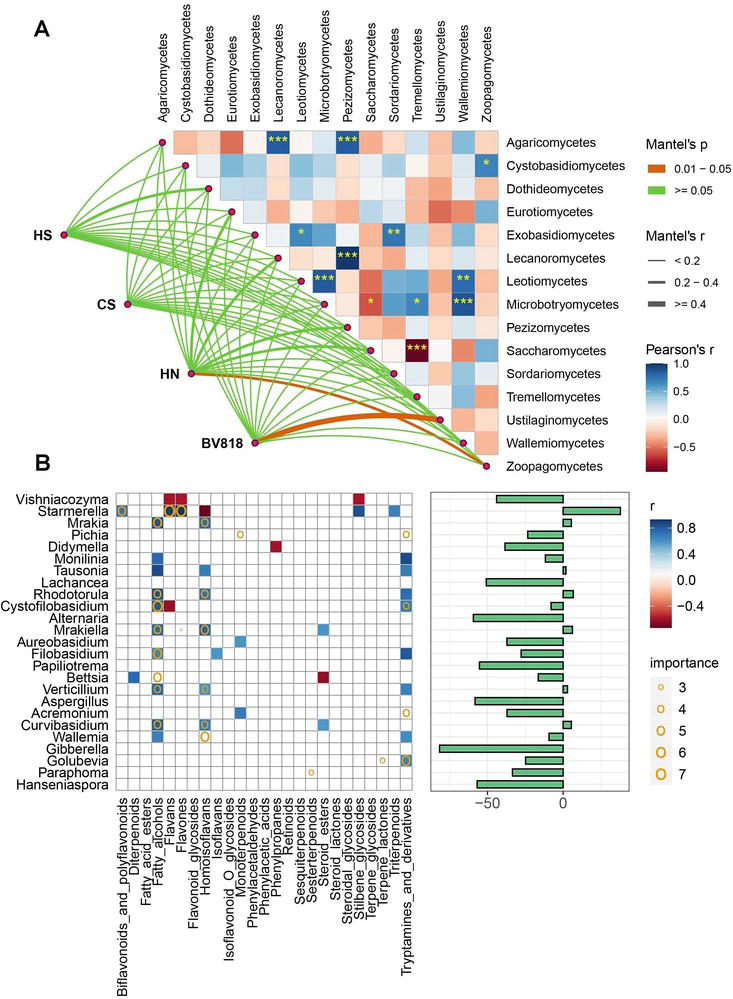
Figure 4.
(A) Correlations of the networked community structures (Bray–Curtis distance) with important fungi orders. The edge width corresponds to Mantel’s r value, and the edge color denotes the statistical significance. Significance levels were labeled as “*” (
The correlation matrix between the functional compounds and the top 25 active microeukaryotic genera was established using a random forest model and Pearson’s correlation coefficients (Figure 4B). Accordingly, many microeukaryotes showed positive correlations. They potentially contributed to the development of terpenoids and flavonoids in icewines, such as
3.3 Characteristic metabolites of V. blanc
One of the reasons why spontaneous fermentation could not be widely applied is that many winemakers treat it as a ‘black-box’, a complex, multi-strains participated system. With the development of systematic biology, the neutral community model (NCM) will be a useful tool to excavate the deep principle of microbial assembly in co-fermentation or spontaneous fermentation [23]. In this study, the NCM predicted a large fraction of the total correlation between the occurrence frequency of ASVs and their relative abundance variations (Figure 5A), with 83%, 73%, 80%, and 76% of the explained microeukaryotic community variations for the BV818, CS, HN, and HS, respectively. The NCM indicates that microeukaryotic communities comply with stochastic assembly instead of deterministic assembly in different fermentation scenarios. Higher R2 suggests the sample is closer to the NCM. Compared to spontaneous fermentation, commercial yeast inoculation (BV818) fits the stochastic process. Moreover, a smaller Nm value suggests more restrictive species dispersal. Therefore, the level of species dispersal in BV818 (Nm = 374) was the lowest in all fermentations, while HS had the maximum species dispersal (Nm = 1005). CS and HN showed similar Nm values. This result is also in accord with the spatial dispersal of microeukaryotic communities during AF; namely, the microeukaryotic communities of HS can evenly develop at three stages of AF, while the ASVs of microeukaryotes of BV818 are mainly concentrated at the end of AF.

Figure 5.
(A) Fit of the neutral community models of fungi communities assembly in the four fermentations. The solid blue lines indicate the best fit to the NCM, and the dashed blue lines represent 95% confidence intervals around the model prediction. OTUs that occurred more or less frequently than predicted by the NCM are shown in different colors (gray dots represent the frequency of occurrence within the 95% confidence interval ranging around the model prediction, orange dots represent above prediction, and purple dots represent below prediction). Nm indicates the metacommunity size times immigration, R2 indicates the fit to this model. Meanwhile, a modified stochasticity ratio was used to assess the relative importance of stochasticity. (B) Coefficients between |β-NTI| and the relative abundance of the six featured metabolites were calculated to evaluate the correlative fitting curves,
β-Nearest taxon index (β-NTI) between samples was calculated to describe the phylogenetic turnover of microeukaryotic communities in the different fermentation scenarios. Linear regression curves between |β-NTI| and the relative abundance of the six characteristic metabolites were constructed (Figure 5B). Among these six characteristic metabolites, β-sitosterol is a plant steroid, empenthrin, gentiopicroside, zerumbone, and qingyangshengenin belong to terpenoids, and phlorizin is a kind of flavonoids. Empenthrin, β-sitosterol, gentiopicroside, phlorizin, and zerumbone positively correlated to |β-NTI| while gentiopicroside and qingyangshengenin showed negative correlations to |β-NTI|. However, β-NTI of all the fermentations were in the range of −2 to 2 (Figure 5C). Although gentiopicroside and qingyangshengenin could affect the stochasticity of microeukaryotic communities, the stochastic process still dominated the phylogenetic turnover of microeukaryotic communities during AF. To assess the relative importance of determinism and stochasticity, modified stochasticity ratio (MST) was carried out to supply the results of NCM (Figure 5D). In four fermentation scenarios, MST of CS (36.1%) was much lower than the boundary (50%), which indicated determinism affected the microeukaryotic assembly of CS. The MST of HS (54.7%) was slightly higher than the MST of HN (53.0%) without a significant difference (
3.4 The specific process of stochastic assembly of microeukaryotic communities and the selection of fermentative specialists
Principal coordinates analysis (PCoA) was carried out to observe the degree of similarity between the microeukaryotic communities of different fermentation scenarios (Figure 6A). The total contribution of the first two components of PCoA can explain 66.11% of variations. The confidence intervals of HN and BV818 have been separated from the spontaneous fermentations (CS and HS). Comparatively, the degree of distribution of BV818 was more concentrated than that of the others while HN and HS showed higher degree of distributions than CS and BV818. The result of PCoA indicated that the microeukaryotic composition of BV818 was different from co-fermentations (HN, CS, and HS), and the concentrated distribution of BV818 also indicated low diversity of microbial participation during AF. Moreover, the top five abundant microeukaryotic genera were selected and located on the PCoA plot. Herein,

Figure 6.
(A) Principal coordinate analysis (PCoA) based on Sørensen dissimilarly displaying differences in community composition of fungal genera in the four icewine fermentations, the top 10 highest active fungi genera were selected and labeled. (B) The relative influence of each community assembly process among four icewine fermentation was defined by the percentage of site pairs governed by each process. (C) Partitioning the turnover and nestedness components of β-diversity of fungi communities in four icewine fermentations. Duncan’s multiple range test was carried out for the significant differences of the model at
Except for the NCM, phylogenetic-bin-based null model analysis was carried out further to infer community assembly mechanisms in the stochastic process (Figure 6B) [20]. The important assembly processes of microeukaryotic communities were heterogeneous selection, homogeneous selection, dispersal limitation, homogenizing dispersal, and drift. Specifically, the assembly process was mainly composed of homogeneous selection (43.1%) and drift (56.2%) in BV818. For HN, dispersal limitation (0.05%), homogeneous selection (45.1%), and drift (49.7%) were the important parts of the stochastic assembly. For CS, the specific stochastic assembly included homogeneous selection (25.5%), dispersal limitation (0.05%), homogenizing dispersal (0.04%), and drift (65.4%). The heterogeneous selection was the characteristic assembly process in HS, which possessed 0.05% of the stochastic assembly. Besides, the proportion of homogeneous selection, dispersal limitation, homogenizing dispersal, and drift was 15.7%, 10.5%, 0.03%, and 65.4% of the stochastic assembly in HS, respectively. In general, drift and homogeneous selection were the major assembly processes during AF of icewine, and HS showed a more complex composition of the assembly process than other fermentations. Partitioning β-diversity of richness was carried out to quantify the result of species replacement between different fermentations (turnover) and species gains or losses between different microeukaryotic communities (nestedness) (Figure 6C). As a result, species replacement played an important role in shaping microeukaryotic communities. The greatest difference between the contributions of β-diversity components was observed in the four fermentation scenarios, where the turnover was about six times higher than nestedness. Specifically, the range of turnover in each fermentation was from 50% to 55% while the range of nestedness was from 5% to 8%. The nestedness of HN was the highest level among the four fermentations. For turnover, BV818 and CS were higher than HN and HS.
To inspect the distribution of OTUs from each fermentation and the specificity of these microeukaryotes during AF, specificity and occupancy were calculated for each OUT, which was then projected onto a plot (SPEC-OCCU plot, Figure 6D). As indicated by the spread of OTUs across occupancy, OTUs from BV818 and HN communities showed highly varied occupancy while the majority of OTUs from CS and HS exhibited more homogenous occupancy, which mainly concentrated at the range of 0–0.25. To find specialist species attributable to each fermentation, we selected OTUs with specificity and occupancy greater or equal to 0.7 (dotted boxes). These microeukaryotes are specific and could be highly fermentative in their fermentation scenarios. The number of these specialist OTUs was BV818 (3 OTUs represent), HN (6 OTUs), CS (2 OTUs), and HS (3 OTUs), respectively. Specifically,
3.5 The analysis of metabolic pathways in four icewine fermentation scenarios
A structural equation modeling (SEM) accounting for the yeast strains, characteristic metabolites, and key assembly factors was built to analyze the interactions of variables in four icewine fermentation scenarios (Figure 7A). Based on the previous results, five yeast strains (

Figure 7.
(A) Structural equation modeling (SEM) accounting for the yeast strains, characteristics, metabolites, and key assembly factors in four icewine fermentation scenarios. (B) Top 20 enriched KEGG pathways of the fungal community in four icewine fermentation scenarios. Numbers adjacent to measured variables are their coefficients with composite variables. Numbers adjacent to arrows are path coefficients are the directly standardized effect size of the relationship. The thickness of the arrow represents the strength of the relationship. Note: CS: spontaneous fermentation using the epiphytic microorganisms from the fully ripe grape berries; HS: spontaneous fermentation using the epiphytic microorganisms from the grape berries in late harvest; HN: icewine fermentation using the self-assembled non-Saccharomyces yeasts group, icewine fermentation using a commercial yeast strain, which belongs to
The top 20 enriched KEGG pathways of four icewine fermentations were shown in Figure 7B, where the rich factors of each KEGG pathway were the ratio of differential genes in this pathway, while the p-value indicated the importance of this pathway in the icewine fermentation. Accordingly, protein digestion, absorption, and ABC transporters were the most important pathways of microeukaryotic metabolism in all fermentation treatments. Compared to commercial yeast, CS, HN, and HS showed similar categories of important KEGG pathways, which also contained mineral absorption, biosynthesis of amino acids, and aminoacyl-tRNA biosynthesis. Lysine degradation was the characteristic pathway for the spontaneous fermentations (CS and HS). Starch and sucrose metabolism were relatively important to HS and BV818, while proximal tubule bicarbonate reclamation was the common pathway in BV818 and CS. Interestingly, valine, leucin, and isoleucine biosynthesis and nicotinate and nicotinamide metabolism were the specific pathways in CS. It indicated more potential volatile metabolites could be synthesized by applying the indigenous yeast community during alcoholic fermentation.
4. Discussion
In this study, the top-down approach was first used to design a self-assembled non-
The spatial dispersal and co-occurrence network of each fermentation scenario performed a dynamic distribution of microeukaryotes and the correlations between microeukaryotic communities during AF. As a result, negative cohesion, such as competition, parasitism, or amensalism, played an important role in the cross-interactions between microeukaryotic communities. The microeukaryotic dispersal of BV818 was mainly concentrated at the end of fermentation, while HN showed the opposite result that the microeukaryotes mainly distributed at the beginning of fermentation. Understandably, most commercial yeasts were
People had been drinking natural wines for thousands of years before sulfur dioxide was applied in the wine industry. Current studies have admitted the importance of a certain strain of commercial yeast in the winemaking industry, while indigenous fungi have been misunderstood as microbial contaminators during AF. In our study, the dominator of AF was indigenous non-
5. Conclusion
This study first explored the assembly process of microeukaryotic communities in four typical icewine fermentation scenarios. Herein, top-down design was carried out to construct a non-
Acknowledgments
We acknowledge Huanren Senpatina Icewine Domaine Co., Ltd. for kindly providing the experimental raw materials.
References
- 1.
Llauradó JM, Rozès N, Constantí M, Mas A. Study of some Saccharomyces cerevisiae strains for winemaking after preadaptation at low temperatures. Journal of Agricultural and Food Chemistry. 2005; 53 :1003-1011 - 2.
Chen K, Zhang L, Qiu S, Wu X, Li J, Ma L. Freeze–thaw cycles characterize varietal aroma of Vidal blanc grape during late harvest by shaping self-assembled microeukaryotic communities. Food Chemistry. 2022; 384 :132553 - 3.
Chen K, Escott C, Loira I, Fresno JMD, Morata A, Tesfaye W, et al. Use of non-Saccharomyces yeasts and oenological tannin in red winemaking: Influence on colour, aroma and sensorial properties of young wines. Food Microbiology. 2018; 69 :51 - 4.
Chen K, Liu C, Wang Y, Wang Z, Li F, Ma L, et al. Predominance of indigenous non-saccharomyces yeasts in the traditional fermentation of greengage wine and their significant contribution to the evolution of terpenes and ethyl esters. Food Research International. 2021; 143 :110253 - 5.
Chen K, Qiu S, Liu C, Zhang L, Wu X, Ma L, et al. Abiotic factors play important roles in complexity and characterization of aroma precursors in Vidal blanc grape. Food Research International. 2022;162 :112015 - 6.
Qiu S, Chen K, Liu C, Wang Y, Chen T, Yan G, et al. Non-saccharomyces yeasts highly contribute to characterisation of flavour profiles in greengage fermentation. Food Research International. 2022; 157 :111391 - 7.
Li R, Lin M, Guo S, Yang S, Han X, Ren M, et al. A fundamental landscape of fungal biogeographical patterns across the main Chinese wine-producing regions and the dominating shaping factors. Food Research International. 2021; 150 :110736 - 8.
Wang QJ, Spence C. Wine complexity: An empirical investigation. Food Quality and Preference. 2018; 68 :238-244 - 9.
Bokulich NA, Thorngate JH, Richardson PM, Mills DA. Microbial biogeography of wine grapes is conditioned by cultivar, vintage, and climate. Proceedings of the National Academy of Sciences. 2014; 111 :E139-E148 - 10.
Chen Y, Zhang W, Yi H, Wang B, Xiao J, Zhou X, et al. Microbial community composition and its role in volatile compound formation during the spontaneous fermentation of ice wine made from Vidal grapes. Process Biochemistry. 2020; 92 :365-377 - 11.
Chen K, Wen J, Ma L, Wen H, Li J. Dynamic changes in norisoprenoids and phenylalanine-derived volatiles in off-vine Vidal blanc grape during late harvest. Food Chemistry. 2019; 289 :645-656 - 12.
Jing W, Min L, Jixin L, Tengzhen M, Shunyu H, Morata A, et al. Chapter 10—Biotechnology of ice wine production. In: Holban AM, Grumezescu AM, editors. Advances in Biotechnology for Food Industry. Academic Press; 2018. pp. 267-300 - 13.
Ma Y, Xu Y, Tang K. Aroma of icewine: A review on how environmental, viticultural, and oenological factors affect the aroma of icewine. Journal of Agricultural and Food Chemistry. 2021; 69 :6943-6957 - 14.
López-Rituerto E, Savorani F, Avenoza A, Busto JH, Peregrina JM, Engelsen SB. Investigations of La Rioja terroir for wine production using 1H NMR metabolomics. Journal of Agricultural and Food Chemistry. 2012; 60 :3452-3461 - 15.
Schrimpe-Rutledge AC, Codreanu SG, Sherrod SD, McLean JA. Untargeted metabolomics strategies—Challenges and emerging directions. Journal of the American Society for Mass Spectrometry. 2016; 27 :1897-1905 - 16.
Patti GJ, Yanes O, Siuzdak G. Metabolomics: The apogee of the omics trilogy. Nature Reviews Molecular Cell Biology. 2012; 13 :263-269 - 17.
Dunn WB, Erban A, Weber RJM, Creek DJ, Brown M, Breitling R, et al. Mass appeal: Metabolite identification in mass spectrometry-focused untargeted metabolomics. Metabolomics. 2013; 9 :44-66 - 18.
Blaženović I, Kind T, Ji J, Fiehn O. Software tools and approaches for compound identification of LC-MS/MS data in metabolomics. Metabolites. 2018; 8 :31 - 19.
Chen Y, Wang H, Wang K, Zhu G, Yang Z, Wang M, et al. Serum metabolomic patterns in patients with aldosterone-producing adenoma. Frontiers in Molecular Biosciences. 2022; 9 :816469 - 20.
Ning D, Yuan M, Wu L, Zhang Y, Guo X, Zhou X, et al. A quantitative framework reveals ecological drivers of grassland microbial community assembly in response to warming. Nature Communications. 2020; 11 :4717 - 21.
Karkaria BD, Fedorec AJH, Barnes CP. Automated design of synthetic microbial communities. Nature Communications. 2021; 12 :672 - 22.
Hernandez DJ, David AS, Menges ES, Searcy CA, Afkhami ME. Environmental stress destabilizes microbial networks. The ISME Journal. 2021; 15 :1722-1734 - 23.
Sloan WT, Lunn M, Woodcock S, Head IM, Nee S, Curtis TP. Quantifying the roles of immigration and chance in shaping prokaryote community structure. Environmental Microbiology. 2006; 8 :732-740 - 24.
Lawson CE, Harcombe WR, Hatzenpichler R, Lindemann SR, Löffler FE, O’Malley MA, et al. Common principles and best practices for engineering microbiomes. Nature Reviews Microbiology. 2019; 17 :725-741 - 25.
Lopez-Toledano A, Mayen M, Merida J, Medina M. Yeasts used to delay browning in white wines. Food Chemistry. 2006; 97 :498-504 - 26.
Martínez C, Cosgaya P, Vásquez C, Gac S, Ganga A. High degree of correlation between molecular polymorphism and geographic origin of wine yeast strains. Journal of Applied Microbiology. 2007; 103 :2185-2195 - 27.
Naumenko ES, Latysheva VT, Gilinsky AM, Rogachev DA, Komarova IN, Salakhutdinov FN, et al. Cardioprotective effect of resveratrol and resveratroloside. Cardiovascular & Hematological Agents in Medicinal Chemistry. 2013; 11 :207-210 - 28.
Zhao X, Tao J, Zhang T, Jiang S, Wei W, Han H, et al. Resveratroloside alleviates postprandial hyperglycemia in diabetic mice by competitively inhibiting α-glucosidase. Journal of Agricultural and Food Chemistry. 2019; 67 :2886-2893 - 29.
Jin Z, Ma J, Zhu G, Zhang H. Discovery of novel anti-cryptosporidial activities from natural products by in vitro high-throughput phenotypic screening. Frontiers in Microbiology. 2019; 10 :1999 - 30.
Zhang H, Wang L, Tan Y, Wang H, Yang F, Chen L, et al. Effect of Pichia on shaping the fermentation microbial community of sauce-flavor baijiu. International Journal of Food Microbiology. 2021; 336 :108898 - 31.
Hollá V, Karkeszová K, Antošová M, Polakovič M. Transglycosylation properties of a Kluyveromyces lactis enzyme preparation: Production of tyrosol β-fructoside using free and immobilized enzyme. Process Biochemistry. 2021; 110 :168-175 - 32.
Barata A, Malfeito-Ferreira M, Loureiro V. The microbial ecology of wine grape berries. International Journal of Food Microbiology. 2012; 153 :243-259 - 33.
Zhang L, Guo K, Wang L, Xu R, Lu D, Zhou Y. Effect of sludge retention time on microbial succession and assembly in thermal hydrolysis pretreated sludge digesters: Deterministic versus stochastic processes. Water Research. 2022; 209 :117900 - 34.
Raimundo e Silva JP, Policarpo IdS, Chaves TP, Coutinho HDM, Alves HdS. A glycosylated β-sitosterol, isolated from Tacinga inamoena (Cactaceae), enhances the antibacterial activity of conventional antibiotics. South African Journal of Botany. 2020; 133 :193-200 - 35.
Hou QZ, Chen DW, Wang YP, Ehmet N, Ma J, Sun K. Analysis of endophyte diversity of Gentiana officinalis among different tissue types and ages and their association with four medicinal secondary metabolites. PeerJ. 2022; 10 :13949 - 36.
Osburn ED, Aylward FO, Barrett JE. Historical land use has long-term effects on microbial community assembly processes in forest soils. ISME Communications. 2021; 1 :48 - 37.
Horvath BO, Sardy DN, Kellner N, Magyar I. Effects of high sugar content on fermentation dynamics and some metabolites of wine-related yeast species Saccharomyces cerevisiae ,S. uvarum andStarmerella bacillaris . Food Technology and Biotechnology. 2020;58 :76-83 - 38.
Li CS, Xu Y, Li LH, Yang XQ , Wang YQ. Acid stress induces cross-protection for cadmium tolerance of multi-stress-tolerant Pichia kudriavzevii by regulating cadmium transport and antioxidant defense system. Journal of Hazardous Materials. 2019;366 :151-159 - 39.
Liu GL, Wang K, Hua MX, Buzdar MA, Chi ZM. Purification and characterization of the cold-active killer toxin from the psychrotolerant yeast Mrakia frigida isolated from sea sediments in Antarctica. Process Biochemistry. 2012;47 :822-827 - 40.
Yang CY, Hao RJ, Du XD, Wang QH, Deng YW, Sun RJ, et al. GC-TOF/MS-based metabolomics studies on the effect of protein sources in formulated diet for pearl oyster Pinctada fucata martensii . Aquaculture. 2018;486 :139-147